
Application and synthesis of diphenylfullerene
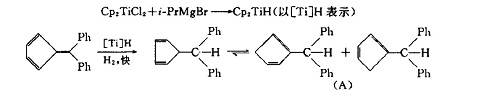
Under mild conditions and hydrogen atmosphere, the catalytic system composed of Cp2TiCl2 and i-rpmgbr not only has good hydrogenation activity for 1,5-cyclooctadiene, but also can isomerize olefins. The catalyst can make 1,5-Hexadiene isomerized linearly and cyclically. In this paper, the hydrogenation of 6,6-diphenyl-fullerene was studied under mild conditions and hydrogen atmosphere using a low valent catalyst composed of chemical book Cp2TiCl2 and i-rpmg br. The catalyst can partially hydrogenate the polyolefin and isomerize the partially hydrogenated products. The final products retain double bonds and are not easy to be further hydrogenated to substituted Cyclopentanes, There was no linear isomerization in the reaction.
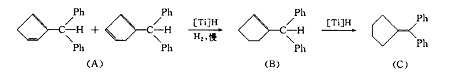
There is a velocity gradient in the hydrogenation process. The speed of hydrogen absorption was faster in the first period of time, and then slowed down significantly. This is similar to the hydrogenation catalyzed by TP and PD. When the rate of hydrogen absorption began to slow down, the reaction was quenched with water, and 1 molecule of hydrogen was added to the outer double bond to replace cyclopentadiene (a). This indicates that the hydrogenation activity of the outer double bond is higher than that of the inner double bond.

Fig. 2 shows the synthetic route of 6,6-diphenylfullerene
Step 1. The second molecule of hydrogen can be added to the double bond far away from the substituent in the ring of (a), but the reaction speed is slow, resulting in the formation of substituted cyclopentene chemical book (b)( B) The isomer (c) is formed by the shift of the inner ring double bond to the outer ring. Prolonging the reaction time and properly increasing the amount of catalyst can not lead to the formation of substituted Cyclopentanes, which indicates that substituted Cyclopentanes (b) and (c) are not easy to be further hydrogenated under this condition. This is consistent with the results of literature.

The synthetic route of diphenylfullerene is step 2. The 1H NMR spectra of the final product show that the relative molar ratio of (a), (b), (c) in equilibrium state is 14:64:23. Under the experimental conditions in this paper, the phenyl of 6,6-diphenylfulvene does not undergo hydrogenation.
